Study Operations Manager
A company is looking for a Study Operations Manager focused on Study Start-Up activities. Key Responsibilities Oversee regional/country/study level implementation of Startup and Site Activation Plans Develop and manage recruitment strategies for assigned studies Coordinate vendor management and ensure timely delivery of study plans Required Qualifications and Education Minimum of 5 years of relevant global/regional clinical trial management experience M.S/M.A/Ph.D or B.A/B.S/nursing degree is essential Experience working at a senior level within Study Start-Up Strong leadership and organizational skills Able to work independently and exercise sound judgment


























































































![Building A Digital PR Strategy: 10 Essential Steps for Beginners [With Examples]](https://buzzsumo.com/wp-content/uploads/2023/09/Building-A-Digital-PR-Strategy-10-Essential-Steps-for-Beginners-With-Examples-bblog-masthead.jpg)



![How One Brand Solved the Marketing Attribution Puzzle [Video]](https://contentmarketinginstitute.com/wp-content/uploads/2025/03/marketing-attribution-model-600x338.png?#)




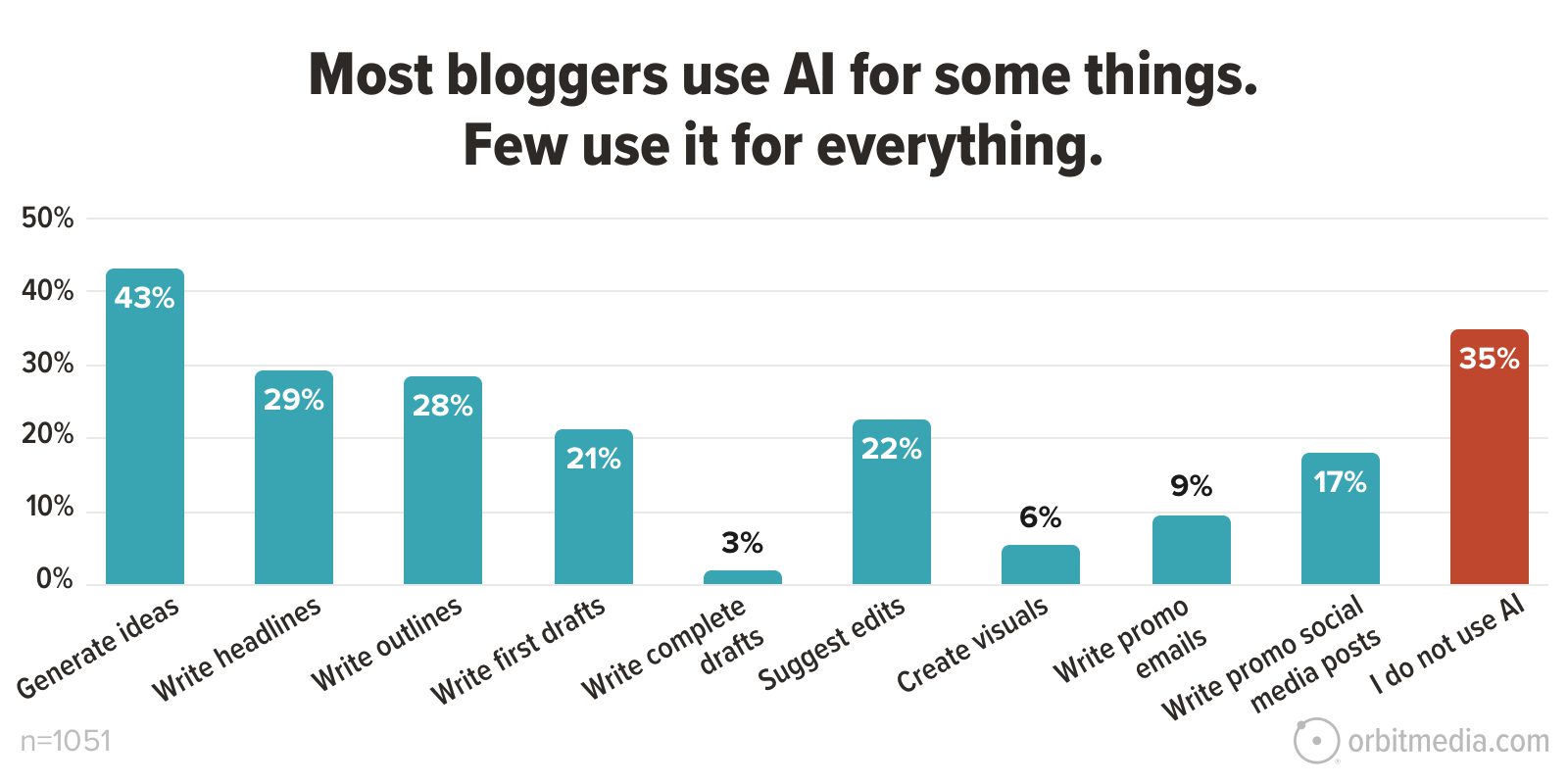
![How to Use GA4 to Track Social Media Traffic: 6 Questions, Answers and Insights [VIDEO]](https://www.orbitmedia.com/wp-content/uploads/2023/06/ab-testing.png)







![[Hybrid] Graphic Designer in Malaysia](https://a5.behance.net/920d3ca46151f30e69b60159b53d15e34fb20338/img/site/generic-share.png)




















