Clinical Research Monitor
A company is looking for a Clinical Research Monitor to support clinical research studies. Key Responsibilities: Monitor protocol and regulatory compliance of clinical research studies across multiple treatment areas Review research data for accuracy and manage documentation and study milestones Write monitoring reports and assist in developing risk monitoring plans and study-related documents Required Qualifications: Bachelor's degree in a relevant area required Minimum of 2+ years of clinical research experience in a complex disease area Prior experience managing clinical trial regulatory documents Preferred clinical research monitoring experience Preferred medical writing experience






































































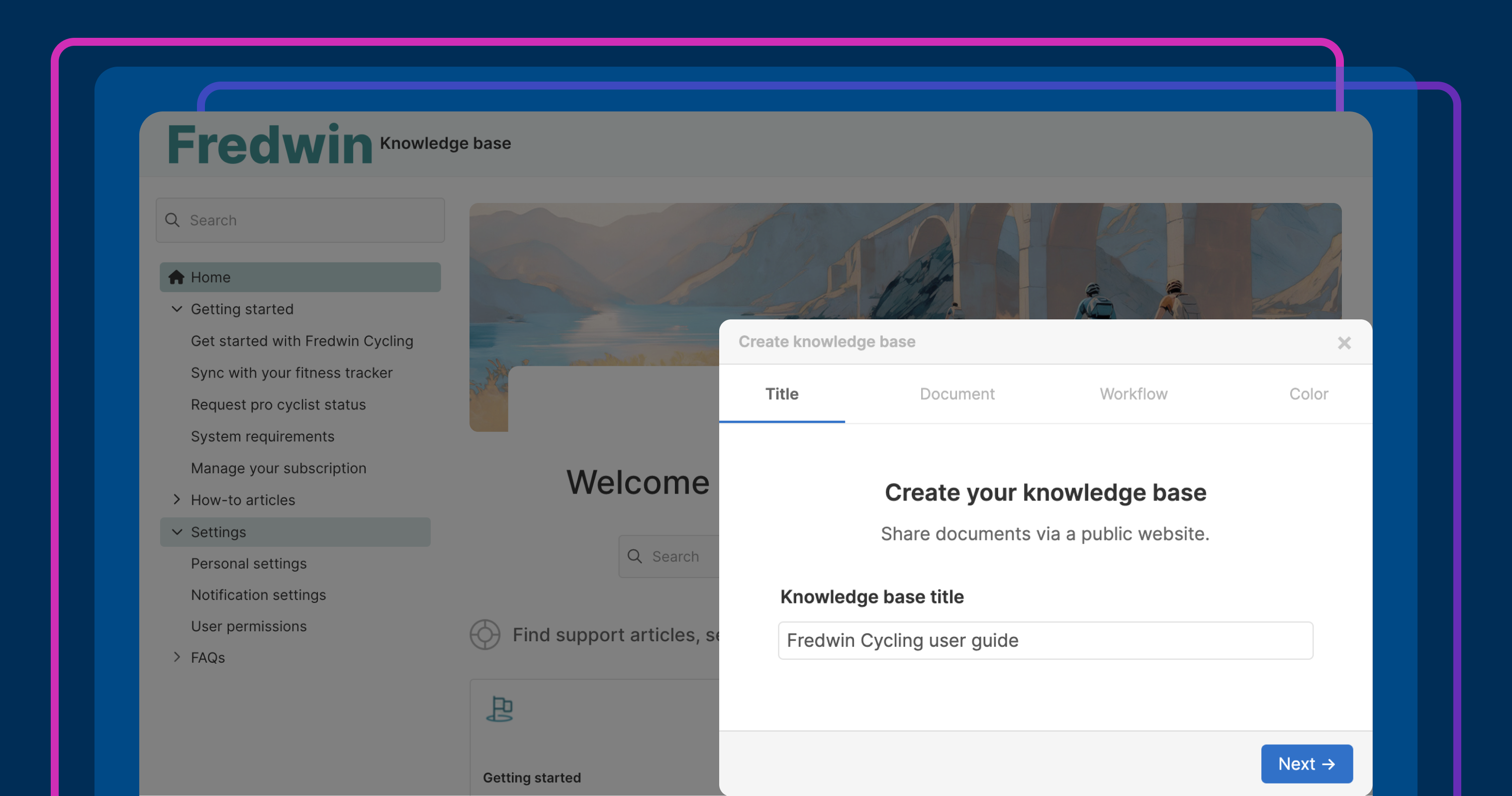






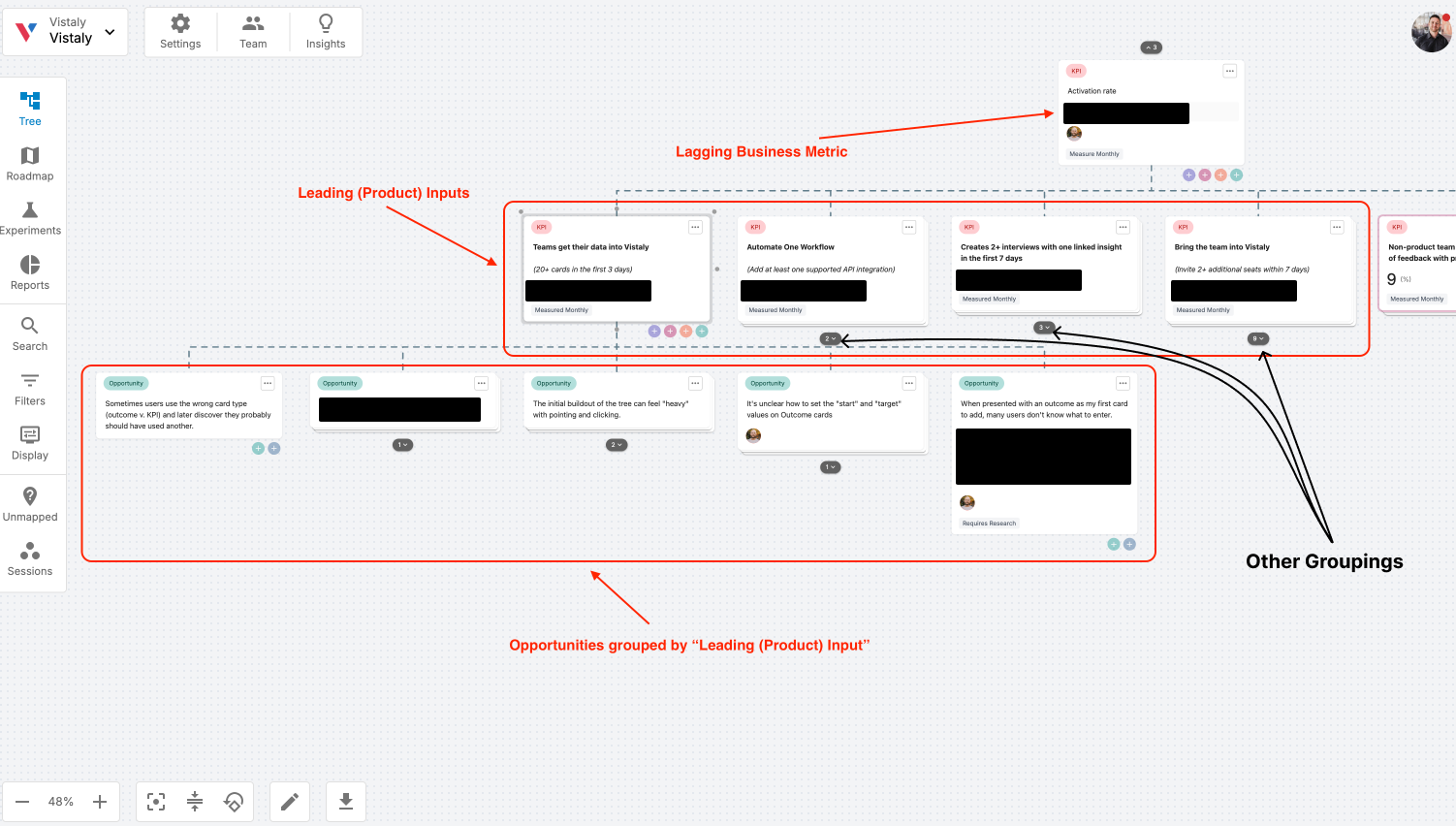
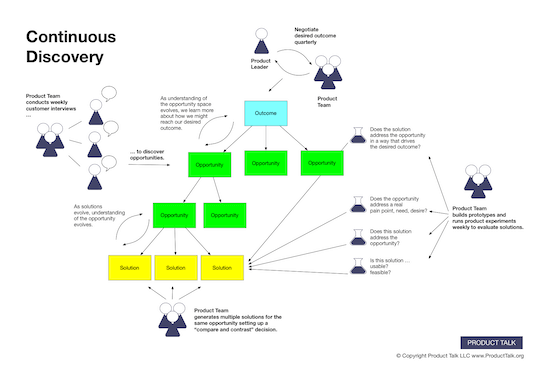














![Building A Digital PR Strategy: 10 Essential Steps for Beginners [With Examples]](https://buzzsumo.com/wp-content/uploads/2023/09/Building-A-Digital-PR-Strategy-10-Essential-Steps-for-Beginners-With-Examples-bblog-masthead.jpg)



![How One Brand Solved the Marketing Attribution Puzzle [Video]](https://contentmarketinginstitute.com/wp-content/uploads/2025/03/marketing-attribution-model-600x338.png?#)




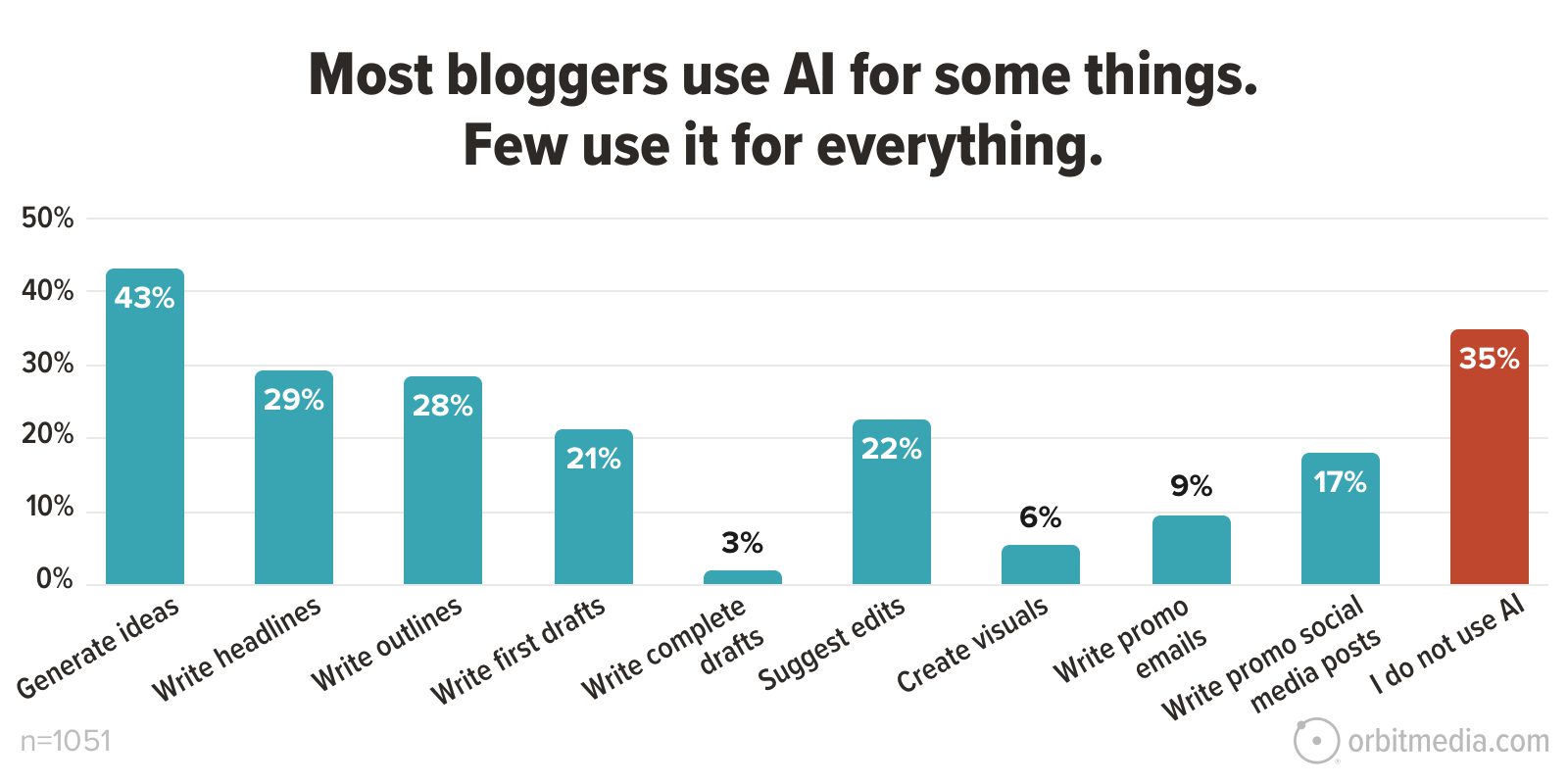
![How to Use GA4 to Track Social Media Traffic: 6 Questions, Answers and Insights [VIDEO]](https://www.orbitmedia.com/wp-content/uploads/2023/06/ab-testing.png)







![[Hybrid] Graphic Designer in Malaysia](https://a5.behance.net/920d3ca46151f30e69b60159b53d15e34fb20338/img/site/generic-share.png)




















